(Chemistry Projects) Class 12th Chemistry Projects for 2009 Exams (DETERMINE THE MASS OF ALUM CRYSTALS)
Disclaimer: This website is NOT associated with CBSE, for official website of CBSE visit - www.cbse.gov.in
Class
12th Chemistry Projects for 2009 Exams
DETERMINE THE MASS OF ALUM CRYSTALS
INTRODUCTION
Aluminium because of its low density, high tensile strength and resistance to corrosion is widely used for the manufacture of airplanes, automobiles, lawn furniture as well as for aluminium cans. Being good conductor of electricity, it is also used for transmission of electricity. Aluminium foil is used for wrapping cigarettes, confectionery items, etc. Aluminium is also used for making utensils. The recycling of aluminium cans and other aluminium products is a very positive contribution to saving our natural resources. Most of the recycled aluminium is melted and recast into other aluminium metal products or used in the production of various aluminium compounds, the most common of which are the alums. Alums are double sulphates having general formula X2SO4.M2(SO4)3.24H2O where,
X = monovalent cation such as Na+, K+, NH4+, etc.
M = trivalent cation such as Al+3, Cr+3, Fe+3, etc.
Some important alums and their names are given below:
Potash Alum: K2SO4.Al2(SO4)3.24H2O
Soda Alum Na2SO4.Al2(SO4)3.24H2O
Chrome Alum K2SO4.Cr2(SO4)3.24H2O
Ferric Alum (NH4)2SO4.Fe2(SO4)3.24H2O
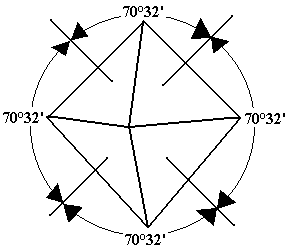
Alums are isomorphous crystalline solids, which are soluble in water. Potash alum is used in paper making, in fire extinguisher, in foodstuffs and in purification of water. Soda alum is used in baking powders and chrome alum is used in tanning leather and waterproofing fabrics. Ferric alum is used in antiseptics. The shape of a potash alum crystal is octahedral.
REQUIREMENTS
Apparatus:
- Conical flasks
- Filter paper
- Piece of aluminium foil
- Burner
- Funnel
Chemicals:
- Potassium Hydroxide (KOH)
- Sulphuric Acid (H2SO4)
- Ethanol (C2H5OH)
THEORY
1. Aluminium metal is treated with hot aqueous KOH solution. Aluminium dissolves as potassium aluminate, KAl(OH)4, salt.
2Al(s) + 2KOH(aq) + 6H2O(l) -> 2KAl(OH)4(aq) + 3H2(g)
2. Potassium aluminate solution on treatment with dil. sulphuric acid first gives ppt. of Al(OH)3, which dissolves on addition of small excess of H2SO4 and heating.
2KAl(OH)4(aq) + H2SO4(aq) -> 2Al(OH)3(s) + K2SO4(aq) + 2H2O(l)
2Al(OH)3(s) + 3H2SO4(aq) -> Al2(SO4)3(aq) + 6H2O(l)
3. The resulting solution is concentrated to near saturation and cooled. On cooling crystals of potash alum crystallize out.
K2SO4(aq) + Al2(SO4)3(aq) + 24H2O(l) ->K2SO4.Al2(SO4)3.24H2O(s)
PROCEDURE
1. Prepare 50ml of 4M KOH solution.
2. Add small pieces of aluminium foil (about 1 gm) in the conical flask containing the KOH solution. Since during this step hydrogen gas is evolved, this step must be done in a well-ventilated area.
3. After all of aluminium has reacted, filter the solution to remove any insoluble impurities.
4. Allow the filtrate to cool. Now add slowly conc. H2SO4 until insoluble Al(OH)3 just forms in the solution
5. Gently heat the mixture until the Al(OH)3 ppt. dissolves. Leave the solution overnight for the crystallization to continue.
6. Take out the crystals and wash them with 50/50 ethanol-water mixture.
7. Determine the mass of the alum crystals.
OBSERVATIONS
Mass of aluminium metal = 1 gm
Mass of potash alum = 13 gm
Colour = White
Shape of crystals =
Octahedral
PRECAUTIONS
1. A few drops of conc. sulphuric acid should be added while preparing saturated solution of aluminium sulphate to prevent its hydrolysis.
2. Aluminium sulphate solution should be clear and not turbid.
3. Cool the conc. solution slowly to get large crystals. Rapid disturbance of solution may change the shape, size and quantity of the crystals.
4. Conc. solution should be cooled undisturbed. A slight disturbance of solution may change the shape, size.